Infodemic and public health professional's response: my crossed experience in Canada and West Africa
Saïdou Sabi Boun

As a health professional trained in Africa, I have been disconcerted to see how rumours can travel rapidly from one continent to another. All this false information in public health now has a name: the infodemic. The WHO defines infodemic as “an overabundance of information, both online and offline,” which is characterized by “deliberate attempts to disseminate wrong information to undermine the public health response and advance alternative agendas of groups or individuals” (WHO, 2020a). Although explanations of infodemic in Africa and Canada are scattered, several myths or issues are common to each continent.
The first and most share misconception is about how speed COVID-19 vaccines have been developed. These vaccines took between 12 and 18 months, whereas it usually takes between 10 and 15 years. The moment spent on the exploratory and preclinical phase has been considerably reduced by using experimental vaccine data from the first two epidemic-causing coronaviruses (SARS-CoV and MERS-CoV). This shrinkage is due to the quick availability of the SARS-CoV-2 genome sequence around January 12, 2020 (WHO, 2020b). This made it possible to skip the exploratory phase and save many months. Therefore, this time saving has allowed some clinical trials to start as early as March 2020, trials designed to reduce time by overlapping clinical trial phases through global mobilization. The first phase I/II trials were quickly followed by a rapid transition to phase III trials as soon as the interim analysis results of phases I and II were available (Kashte et al., 2021; Ndwandwe et Wiysonge, 2021).
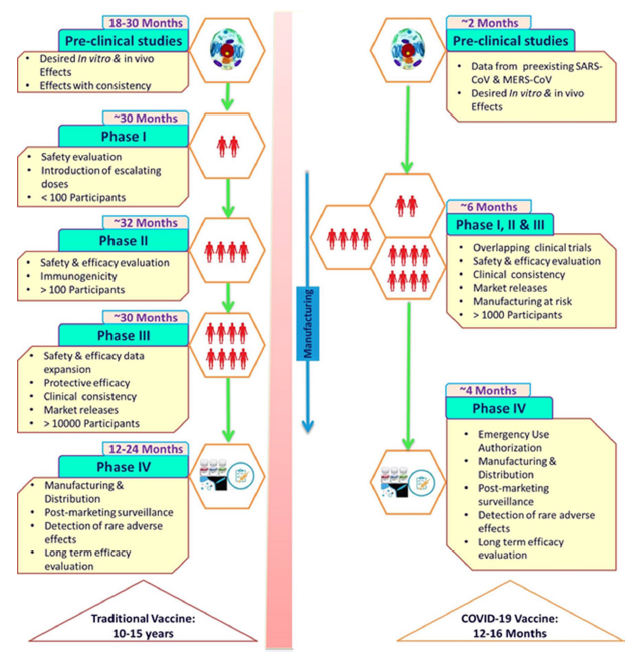
Fig. 1 Rapid development of COVID-19 vaccine as compared to traditional vaccine development (Kashte et al., 2021)
The second myth doubts the equivalence of COVISHIELD produced in India to AstraZeneca’s VAXZEVRIA developed in England. In one of her videos, a pan-Africanist activist, Nathalie Yamb, who has a large obeying on social media, mentioned the difficulty for people immunized in Africa to have their vaccination accepted in Western countries. In this video, she invited her listeners to refuse COVID-19 vaccines. She said, referring to the COVISHIELD, “They have developed a vaccine for Europe and a vaccine for Africa under the same brand name. And today, for travel to Europe, they recognize the version for whites, but not the version for Negroes” (Jeune Afrique, 2021). This pan-Africanist rhetoric has resonated with the African population, particularly the African diaspora in Europe and Canada, reinforcing vaccine hesitancy. The COVISHIELD vaccine results from a voluntary license sharing between AstraZeneca and the Indian laboratory Serum Institute of India to increase the availability of vaccines and thus facilitate their access to the most significant number (WHO, 2020b). Besides being approved by the WHO, the vaccine was approved in some Western countries, notably in Canada, where it was administered to thousands of people, including the Québec Minister of Health, Christian Dubé, before being withdrawn due to the appearance of adverse effects (Radio Canada, 2021).
The last myth is probably the most logically structured. It is the freedom to dispose of one’s body and be free to be vaccinated or not. On the face of it, vaccination in most African countries and Canada is not mandatory, even if it is compulsory. Are individual rights being curtailed by strongly encouraging the population to vaccinate? Public health has three characteristics: initiatives come from the health professional, not the patient, and the target of public health interventions are population-based. As a result, they may have no significant effect on individuals or be harmful to some. This is the prevention paradox. Finally, public health interventions are omnipresent, so it is difficult for individuals to avoid them. These three (3) specificities of public health led to the possibility of moral conflict (MacDonald, 2014; MacDonald, 2015). Frameworks are then used to implement interventions to resolve the ethical conflicts encountered. Several schemes depend on the political context and the situation, but the one of interest to us is the Schröder-Bäck framework, which includes seven (7) principles: 1) non-maleficence: does the COVID-19 vaccine harm, anyone? 2) Beneficence: is the vaccine beneficial for all populations? 3) Health maximization: has the vaccine been proven to be effective? Is there any evidence that the vaccine improves health? 4) Efficiency: Will the vaccine be cost-effective? Or is it better to use another, more efficient intervention? 5) Autonomy: Is vaccination coercive? Does it break on fundamental rights? 6) Justice: Will vaccination cause stigma? Will adverse effects of the intervention be adequately addressed? 7) Proportionality: Is the burden imposed on some individuals more minor than the collective goods obtained? When referring autonomy to dispose of one’s body, do not forget to include the other six (6) principles in the deliberation (Schröder-Bäck et al., 2014).
Finally, I would like to draw attention to the difference between scientific controversies and infodemic. Although the line may seem thin, scientific controversy is intrinsic to science. It is the permanent questioning of scientific truths.
References
- D. Ndwandwe et C. Wiysonge. (2021). COVID-19 vaccines. Current Opinion in Immunology 2021, 71:111—116.
- Jeune Afrique. (2021). Covid-19 : pourquoi les Africains sont-ils victimes d’un apartheid vaccinal.
- MacDonald, M. (2014). Introduction to public health ethics 1: Background. Montréal, Québec: National Collaborating Centre for Healthy Public Policy.
- MacDonald, M. (2015). Introduction to public health ethics 3: Frameworks for public health ethics. Montréal, Québec: National Collaborating Centre for Healthy Public Policy.
- RadioCanada. (18 mars 2021). Le ministre Christian Dubé a reçu le vaccin d’AstraZeneca.
- S. Kashte, A. Gulbake, S. F. El‑Amin et A. Gupta. (2021). COVID‑19 vaccines: rapid development, implications, challenges and future prospect. Human Cell (2021) 34:711—733
- Schröder-Bäck et al. (2014). Teaching seven principles for public health ethics: towards a curriculum for a short course on ethics in public health programmes. BMC Medical Ethics 2014, 15:73
- WHO. (2020a). Managing the COVID-19 infodemic: Promoting healthy behaviours and mitigating the harm from misinformation and disinformation.
- WHO. (2020b). Listings of WHO’s response to COVID-19.
- WHO. (2020c). 172 countries and multiple candidate vaccines engaged in COVID-19 vaccine Global Access Facility.
Saïdou Sabi Boun1,2
1M. Sc Public Health; Université de Montréal, saidou.sabi.boun@umontreal.ca
2M. Sc Management DIAH; Université de Laval, saidou.sabi-boun.1@ulaval.ca
